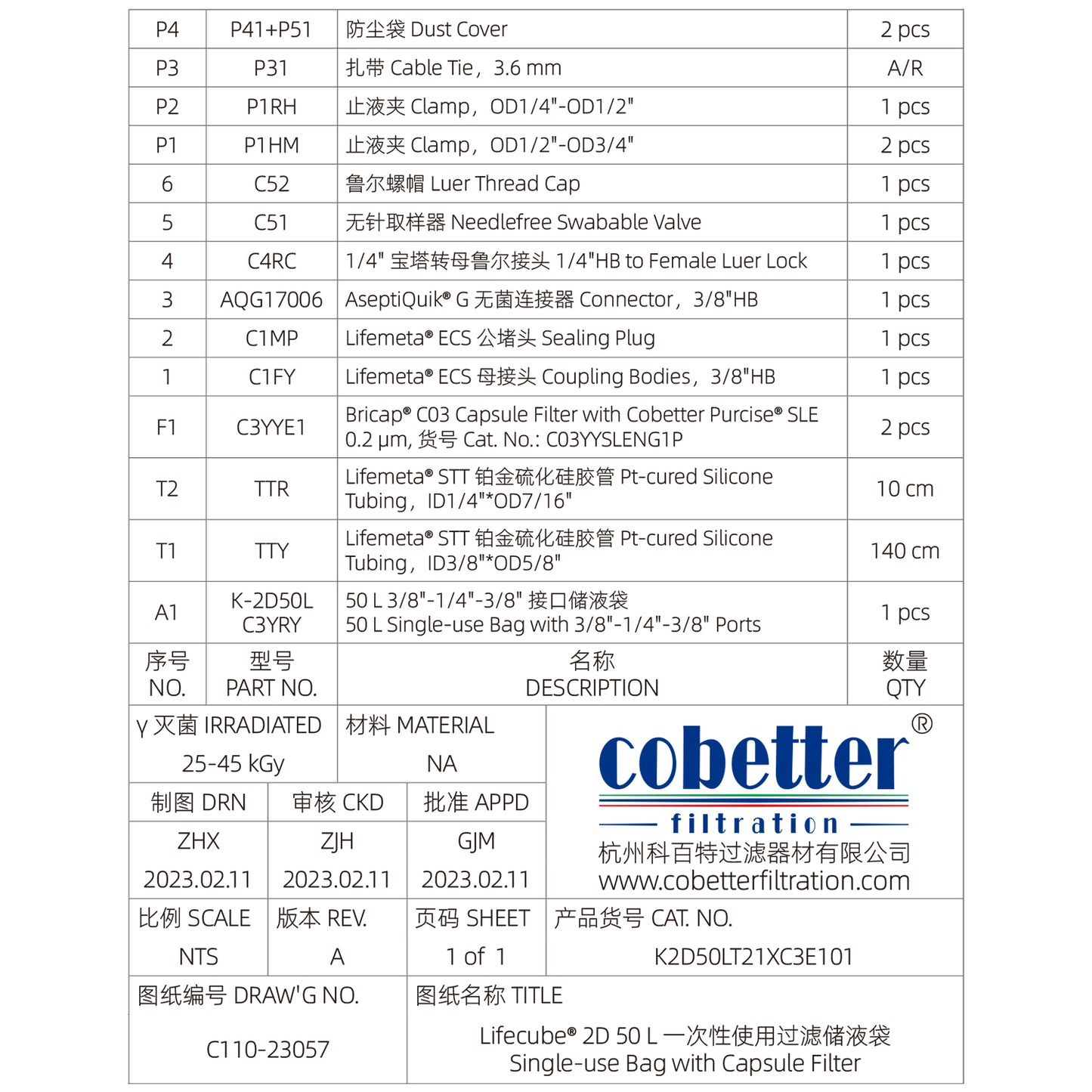
Lifecube™ single-use 2D bag assemblies with filter are sterile, pre-assembled systems designed for fluid handling in biopharmaceutical and life sciences applications. The single-use bag assemblies integrate a filtration unit, ensuring aseptic processing while eliminating cross-contamination risks. For tailored solutions, explore our Custom Single-use Assemblies.
Bag Assemblies with Topene™ Film
Cobetter Topene RB blown film designed for biological processes is composed of ULDPE (ultra-low density polyethylene), EVA (ethylene-acetic acid ethylene copolymer), EVOH (ethylene-vinyl alcohol copolymer) and LDPE (low density polyethylene). Cobetter Lifecube™ 2D bioprocess bags are available in 1, 2, 5, 10, 20, 35 and 50 L sizes.
Tubing
Available in both Pt-cured Silicone and TPE (thermoplastic elastomer) tubing.
Inlet and outlet Connectors
Multiple types and sizes of connectors are available, including ECS Connector, Aseptic Connector, TC, and Plug.
Sampling Port
Needle-free swabable valve, sampling plug, male and female Lure Lock.
Capsule Filter
Cobetter Bricap™ C / L / T series sterilizing-grade filters.
Cobetter Bag Assemblies Advantages:
- Reduced Contamination Risk: Pre-sterilized and ready-to-use, eliminating cleaning validation
- Cost-Efficient: Lower cost (no CIP/SIP systems) and reduced downtime
- Scalability: from R&D to commercial production
Regulatory Compliance: Meets cGMP, USP Class VI, and FDA standards. - Flexibility: Compatible with single-use bioreactors and downstream equipment.
Regulatory Compliance
- ISO 9001:2015 quality management system
- ISO Class 4.8 / ISO 7 cleanroom production
- ADCF raw materials, complies with FDA 21 CFR Part 177-182(Indirect food additives)
- Meet the requirement of current USP <87> Class VI, Biological Reactivity Test, In Vitro
- Meet the criteria of current USP <88> Class VI, Biological Reactivity Test, In Vivo
- Aqueous extraction contains <0.25 EU/mL as determined by Limulus Amebocyte Lysate (LAL), USP <85>
- Particulate matter in the product eluent meets the requirements in USP <788> for large volume parenterals
- Verified gamma irradiation dose according to ISO® 11137