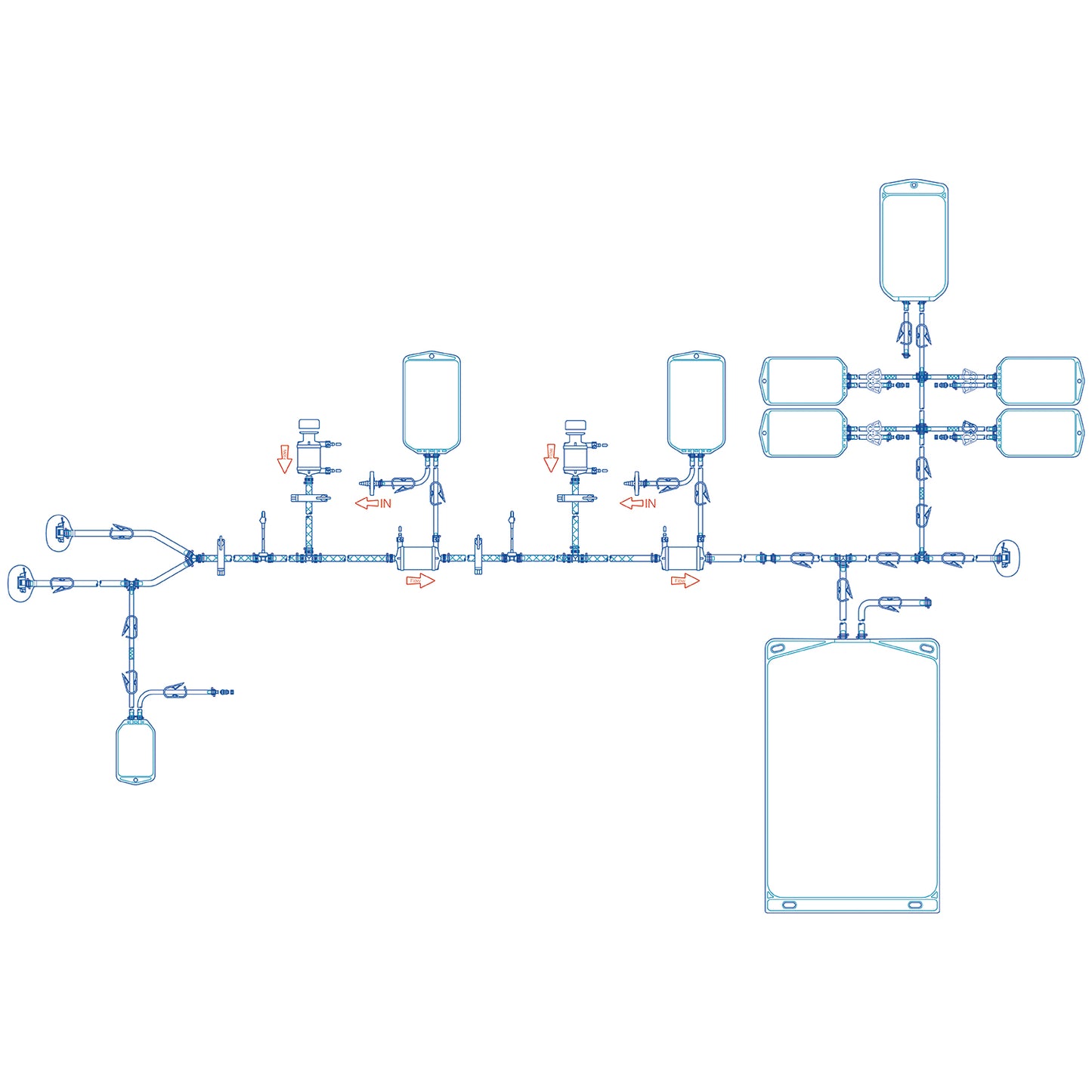
Lifecube™ Single-Use PUPSIT Assembly
*Not for medical use
Couldn't load pickup availability
| Physical Properties | Product Name | Lifecube™ PUPSIT |
| Sterilizing-grade Filter | Bricap™ C03 Capsule Filter 0.2 μm PES membrane, EFA 660cm² | |
| Gas Filter | Bricap™ C02 Capsule Filter 0.2μm PTFE membrane, EFA 640cm² | |
| Tubing | Pt-Cured Silicone | |
| Volume of Single-Use Bag | 500 mL/ 5L | |
| Applications | Typical Application | Terminal Sterilization Filtration (EU GMP requirements) |
| Customized Solutions | Configuration-to-Order (CTO): Short delivery time Engineer-To-Order (ETO) : Flexible design |
|
| Regulatory Compliance | Compliance | USP <788> Particulate Matter USP <87> Cytotoxicity USP <88> Biological Reactivity FDA Indirect Food Additives, 21 CFR 177-182 ADCF Materials |
| Bacterial Endotoxin | Endotoxin Limit of WFI System<0.25 EU/mL with the Limulus Amebocyte Lysate (LAL), USP <85> | |
| Sterility | Delivery Condition | Sterile |
| Sterilization Method | Gamma irradiation at 25–45 kGy | |
| Package | Pack Size | Pack of 1 |
| Packaging | Individually packed in double PE bag |