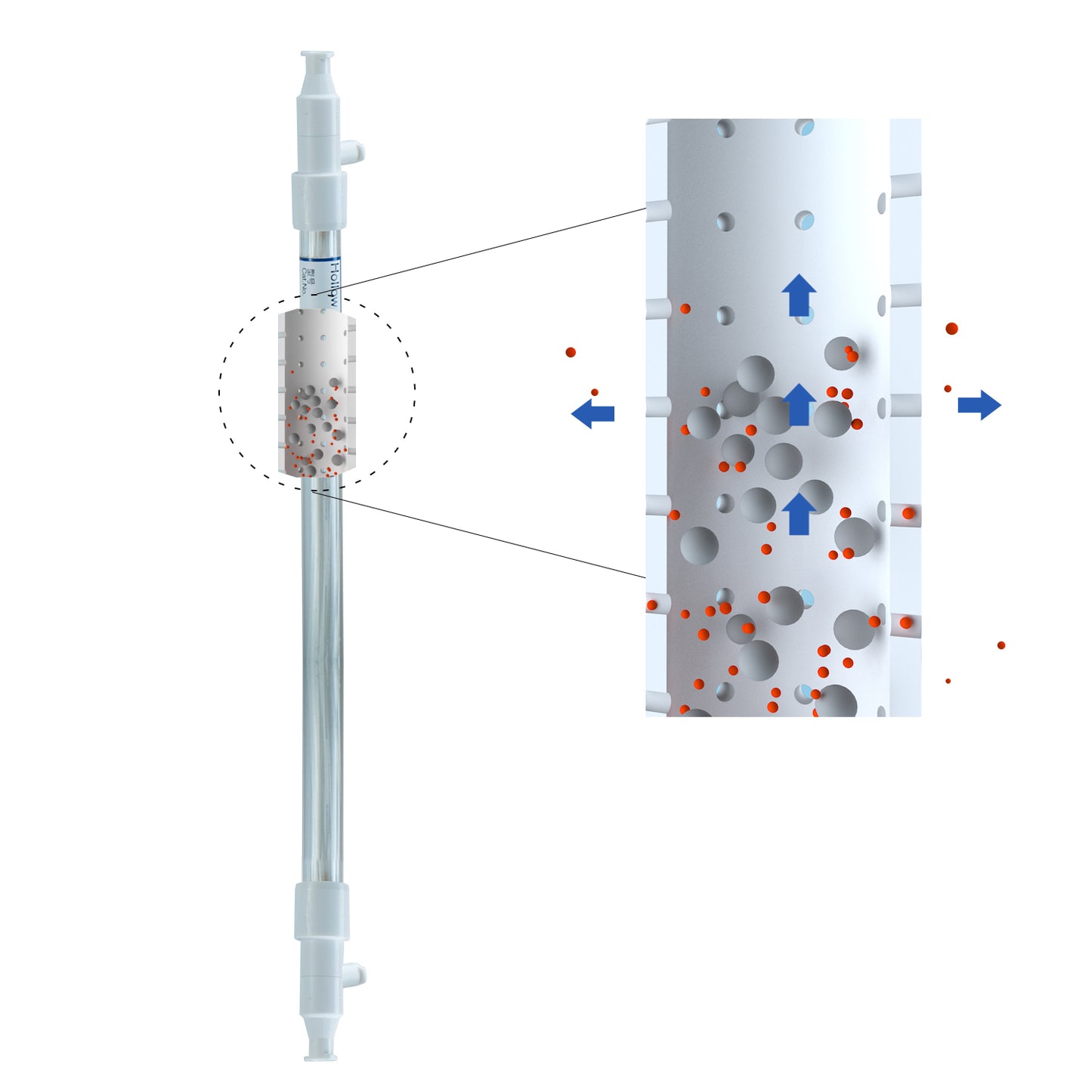
Biophsep™ Mini Hollow Fiber Filter mPES Membrane, Microfiltration, Reusable
Regular price
$85.00 USD
Couldn't load pickup availability
*Ships within 9-12 Business Days
*Customers are responsible for any import duties or taxes
*Read our
Shipping Policy
and
Refund Policy
*Not for medical use
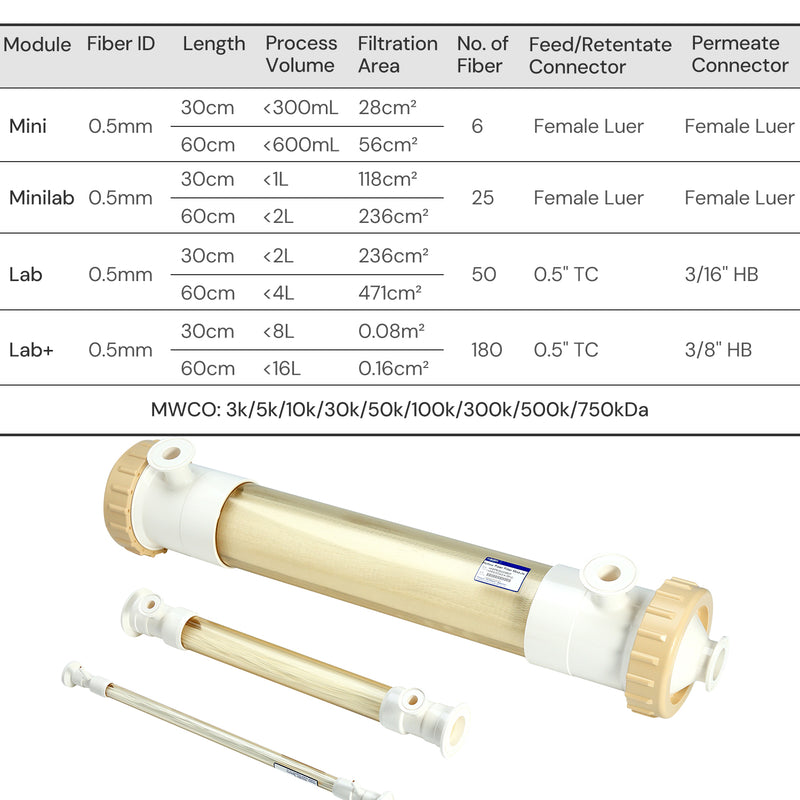
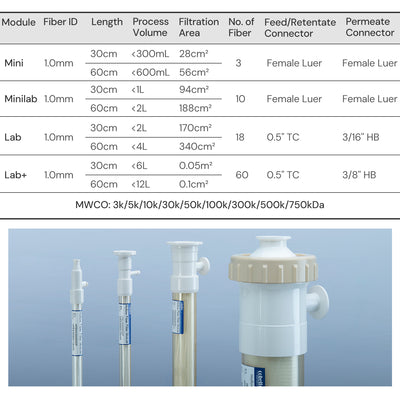
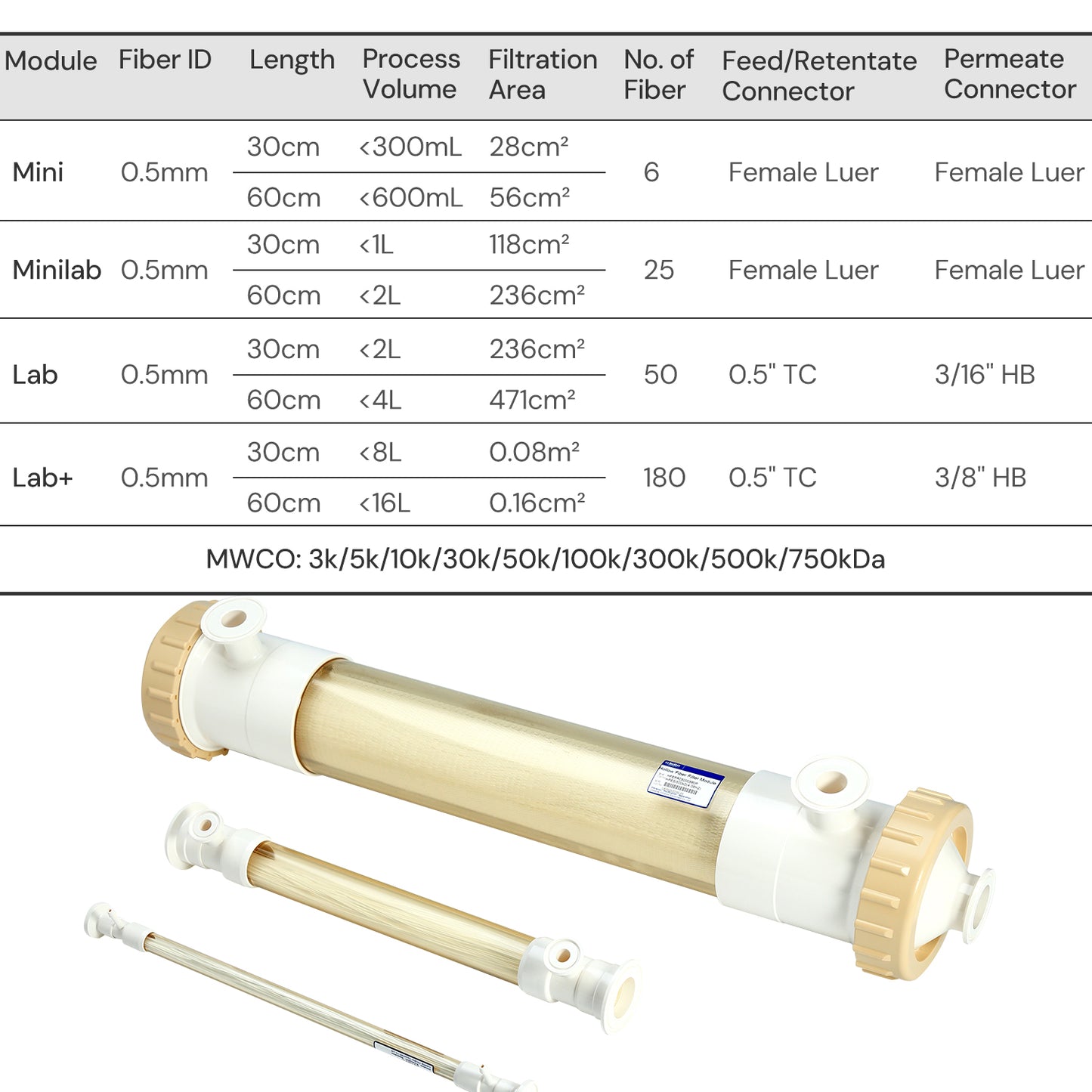
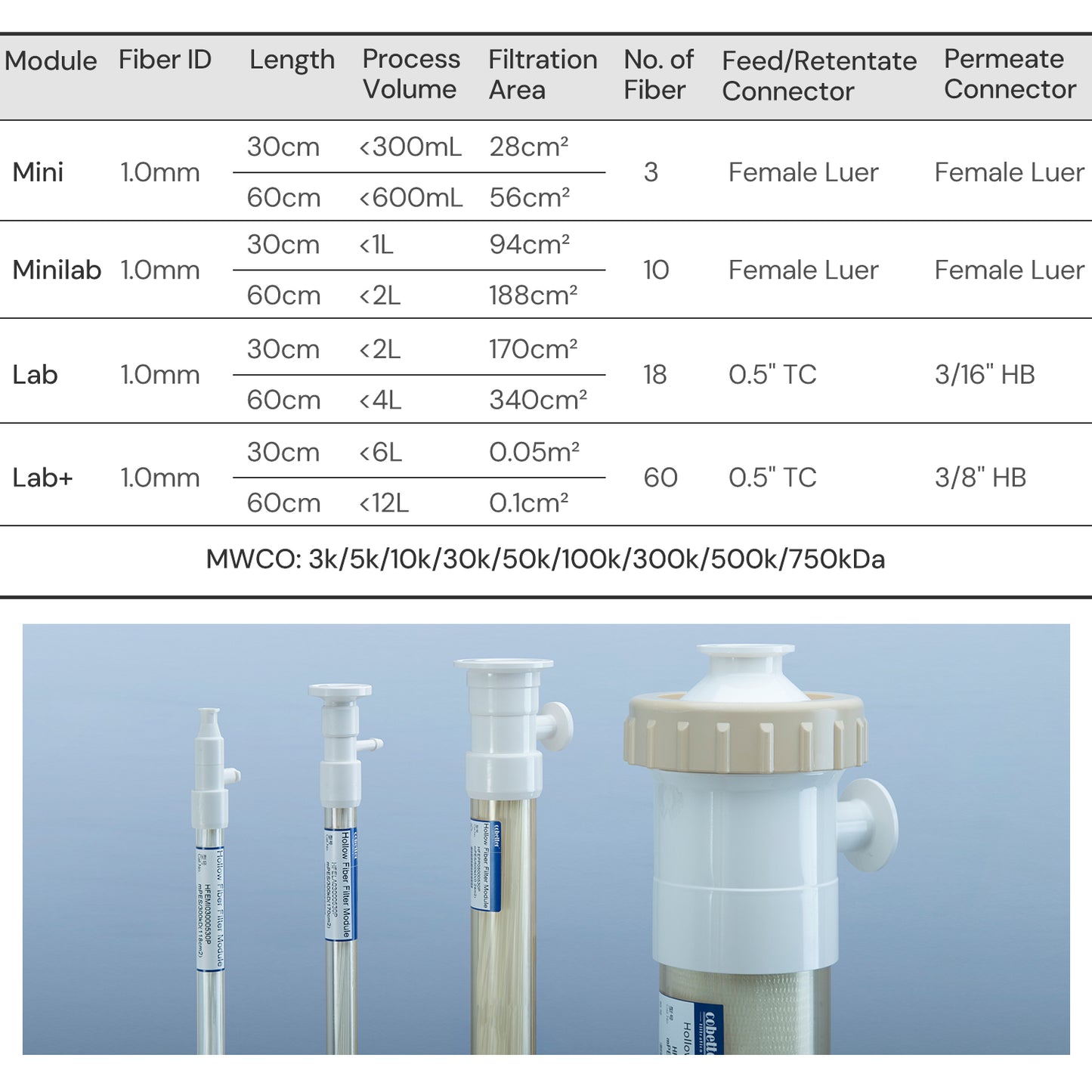
| Physical Properties | Filter Format | Mini Hollow Fiber |
| Scale | Lab Scale, Process Development | |
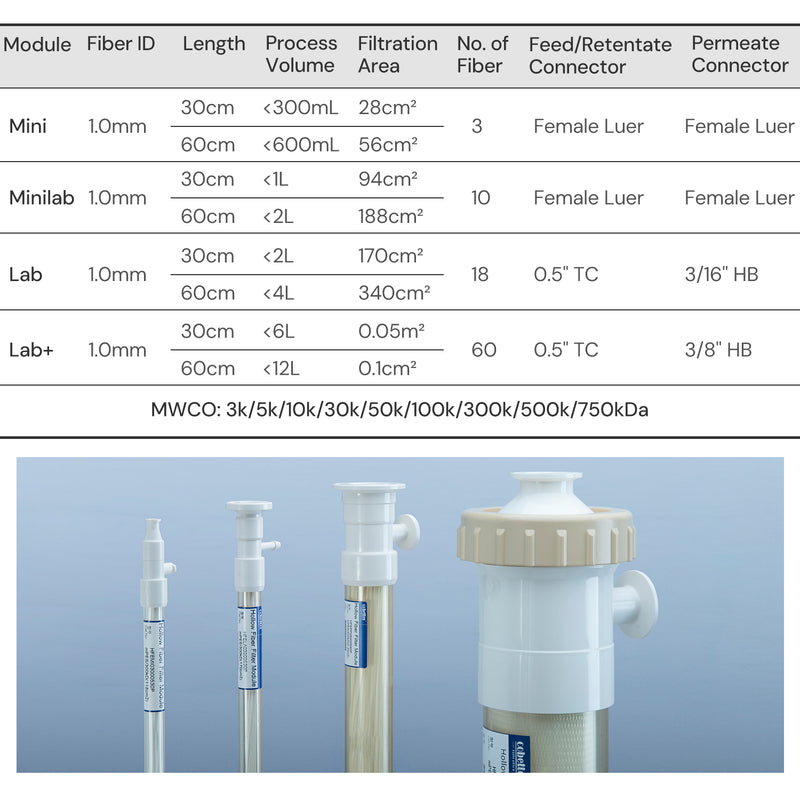
| Fiber ID | 1.0 mm | |
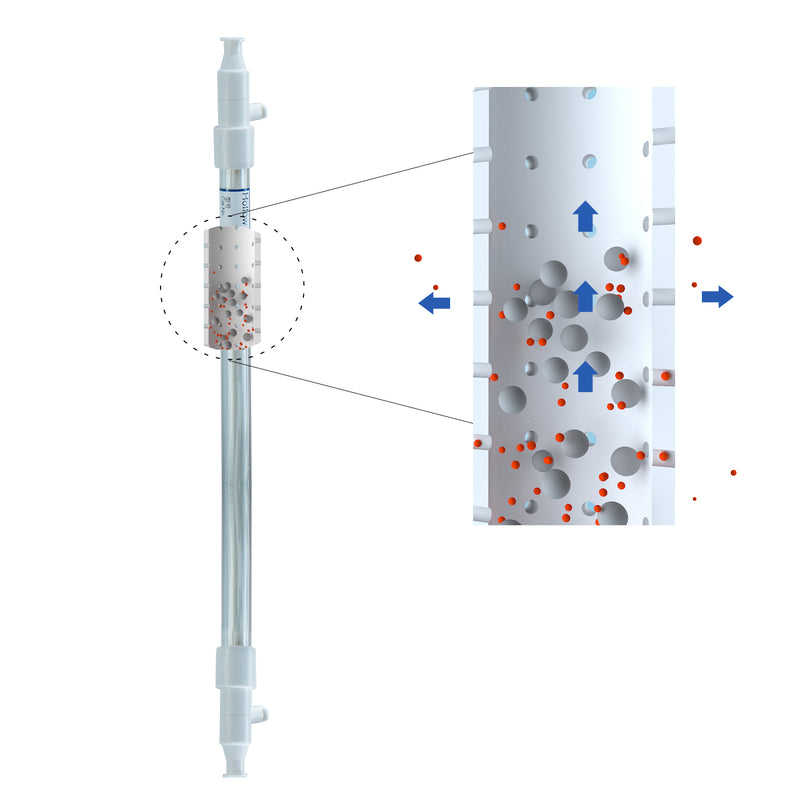
| Flow Path |
30 mm (ODxLength: 1 * 34.6 cm) 60 mm (ODxLength: 1 * 64.6 cm) |
|
| MWCO | 0.1/ 0.2/ 0.45/ 0.65 μm | |
| Applications | Typical Application |
E. coli concentration, E. coli homogenate clarification (750 kDa / 0.22 μm) Insulin protein crystallization (0.45 μm) LNP/Exosome concentration and clarification (100/ 300 kDa) |
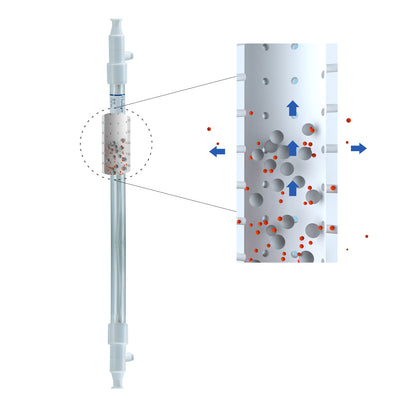
| Materials | Membrane Material | Modified Polyethersulfone (mPES) |
| Housing Material | Polysulfone | |
| Seals (internal) | Epoxy Resin | |
| Ports | Feed/Retentate Port | Female Luer Lock |
| Permeate Port | Female Luer lock | |
| Regulatory Compliance | Compliance | USP <788> Particulate Matter USP <87> Cytotoxicity USP <88> Biological Reactivity FDA Indirect Food Additives, 21 CFR 177-182 ADCF Materials |
| Endotoxin | <0.25 EU/mL bacterial endotoxins (LAL test, Aqueous extraction), USP<85> | |
| Usage Protocols | Reusable | Yes |
| Pre-use Treatment | NaOH Sanitization for Pyrogen Removal | |
| Sterility | Non-sterile | |
| Package | Pack Size | Pack of 1 |
| Packaging | Individually packed in a double PE bag |
Key Operating Parameters
| Fiber ID | Length | EFA | Process Volume | Number of Fibers | Flow Rate @2000s-1 (mL/min) | Flow Rate @6000s-1 (mL/min) |
| 1.0 mm | 30 cm | 28 cm² | < 140 mL | 3 | 35 | 106 |
| 1.0 mm | 60 cm | 56 cm² | < 280 mL | 3 | 35 | 106 |
Choose Fiber ID
- Module with fiber of 0.5mm ID is widely used in most application scenarios to improve mass transfer efficiency.
- Module with fiber of 1.0mm ID is ideal for products with high cell density or high solid content or high viscosity.
- Only module with fiber of 1.0mm ID is autoclavable, as steam cannot enter the fiber of 0.5mm ID effectively.